 We will use E instead of NO 2 as a general mechanism: Let’s why the nitration of nitrobenzene occurs mainly at the meta position:Īs usual, go ahead and draw the complete mechanism with the resonance structures. There is a separate post about this which you can check here. The only exceptions are the halogens that deactivate the aromatic ring yet directing the electrophile to the ortho and para positions. And the larger the groups, the higher the strain, therefore the higher the regioselectivity:įor even larger groups it is possible that no substitution will occur at the ortho position.ĭeactivating groups slow down electrophilic aromatic substitutions and direct the substituents to the meta position. In general, the para product is more favorable because of the steric strain between the closer located ortho substituents. In addition, the ortho and para are not formed in an equal amount either. This is a significant stabilization of the transition state since all the atoms have complete octets (check the stability of resonance structures). To explain the regioselectivity, draw all the resonance structures of the sigma complex:Īnd what we see is that in this case, the ortho and para substitutions have an extra resonance structure involving the lone pair of the oxygen. In addition, halogenations are the fastest among all the electrophilic aromatic substitution reactions: For example, anisole is an activated aromatic ring and does not even require a catalyst to be halogenated because the resonance effect is very strong. 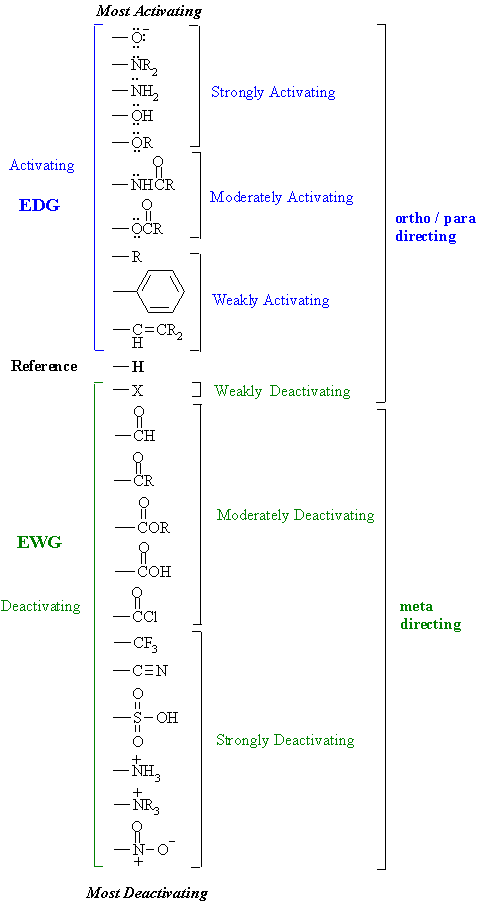
Let’s now look at an example where the aromatic ring is activated by the resonance effect. They all stabilize the transition state, make the reaction faster directing the electrophile to the ortho, parapositions. This resonance contributor stabilizes the transition state thus making the ortho and para substitution faster than meta substitution.Īnd this is true for any activating group. 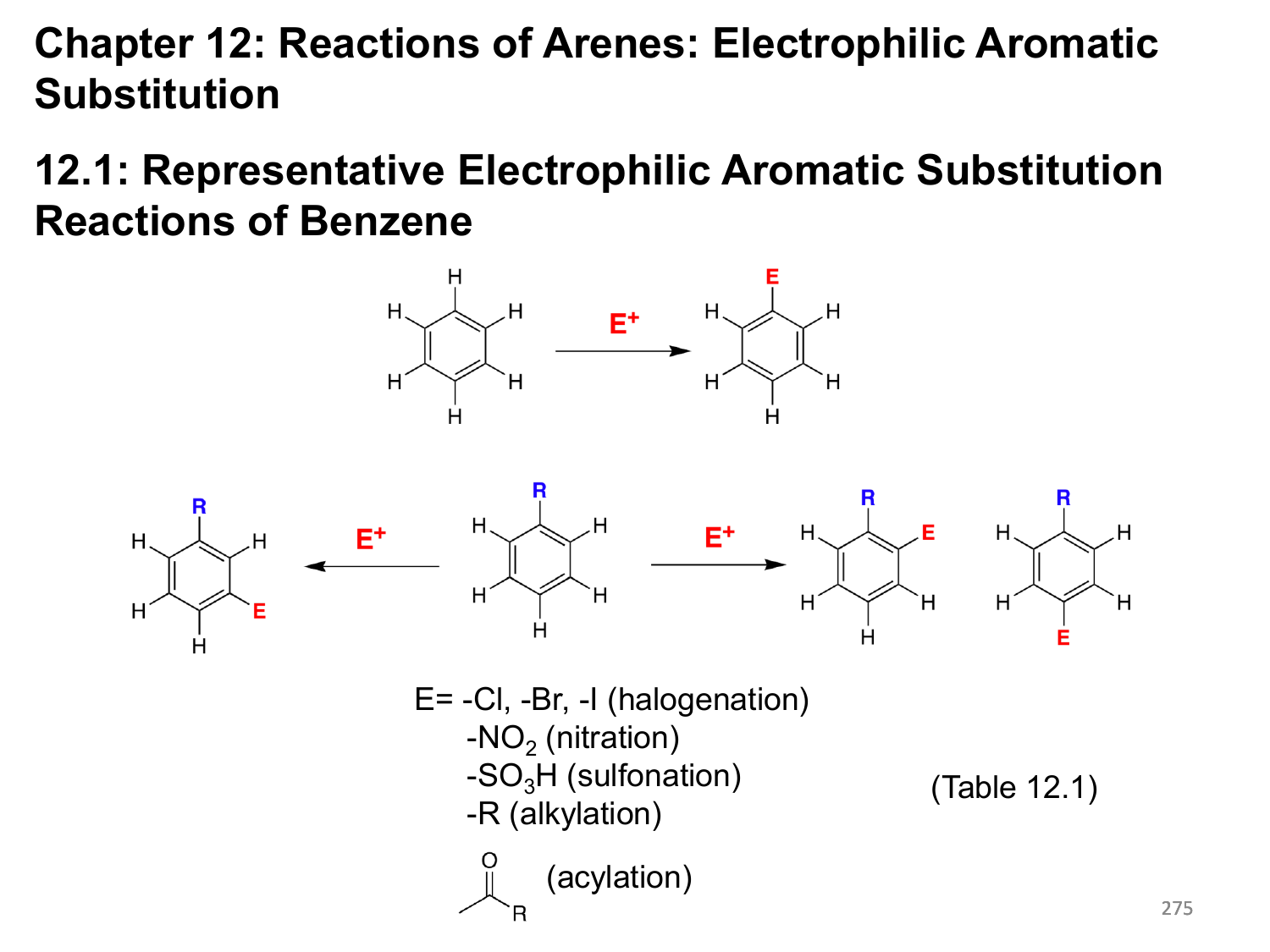
In both ortho and para substitutions, there is a resonance structure with a tertiary carbocation stabilized by stronger hyperconjugation. To explain this regioselectivity, we need to draw the complete electrophilic substitution mechanism including all the resonance strucutures of the sigma complex:ĭo you use any advantage of the ortho and para substituted resonance structures? Think about the stability of carbocations In toluene, the ring is activated by an inductive effect since the methyl an electron-donating group and electrophilic aromatic substitutions of toluene produce the ortho and para products in great excess compared to the meta product: For now, let’s see how the activation and ortho, para regioselectivity works by the inductive and resonance effects. You can check this post for more details about the activating and deactivating groups. The following table summarizes the activating and deactivating groups and their directing effect in electrophilic aromatic substitution reactions: If the atom connected to the aromatic ring has a alone pair of electrons, then the ring is activated by the resonance effect which is generally a stronger contributor: 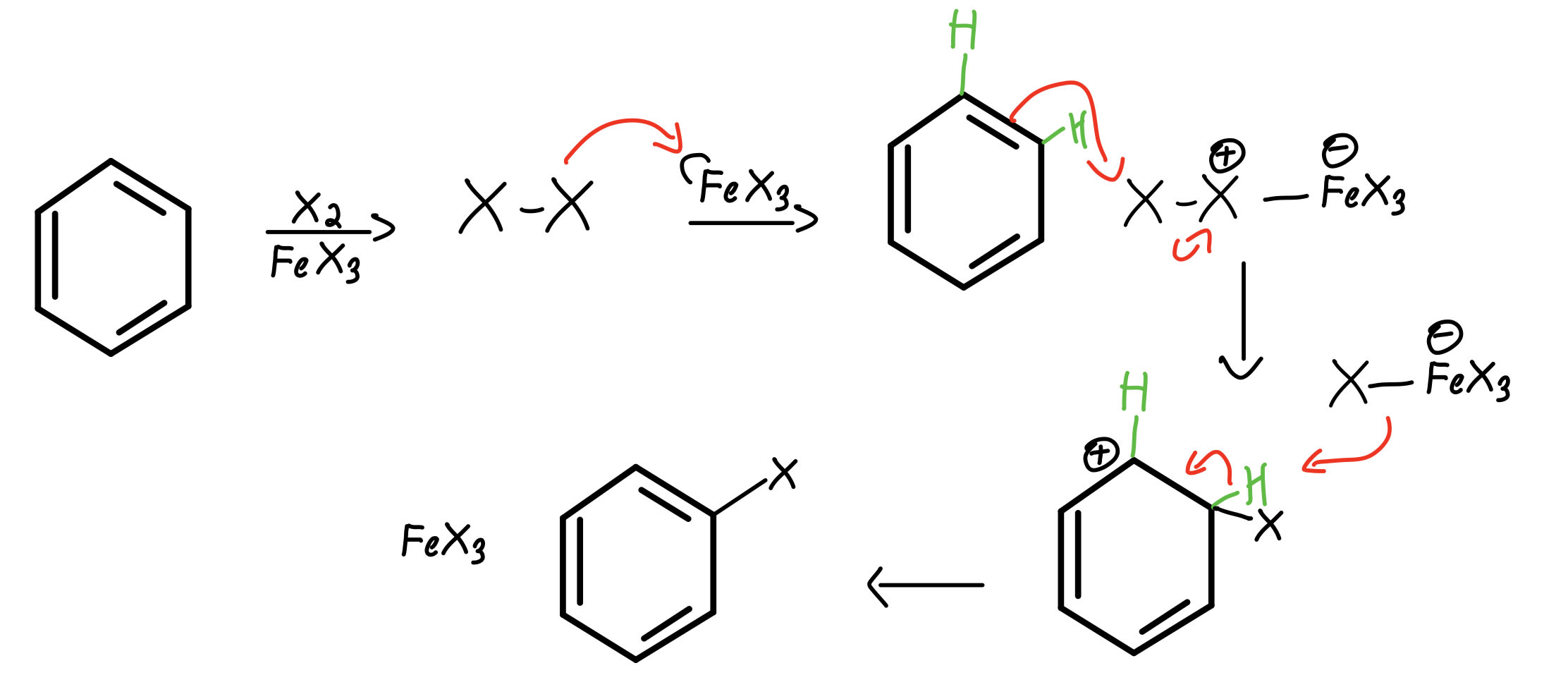
Halogens, on the other hand are more electronegative than carbon and when connected to the ring, they pull the electron density by the inductive effect and thus reduce its reactivity. And, in general, any alkyl group is an activator. For example, a methyl group activates the ring since the carbon is connected to three hydrogens and being more electronegative it pulls the electron density and donates to the ring. There can be electron-donating (activating) and electron withdrawing (deactivating) groups. The inductive effect is a result of different electronegativities of the carbon in the ring and the atom connected to it. The activation and deactivation of the aromatic ring are caused by inductive or resonance effects (or both). Here is the short answer to ‘how do I know if the electrophile will go to ortho, para or meta position?’Īny Activating group directs the electrophile to the orthoand parapositions.Īny deactivating group directs the electrophile to the metaposition.Īn activated ring means it undergoes an electrophilic aromatic substitution faster than benzene and deactivated rings react slower than benzene. As a reminder, the ortho-, meta and para are the relative positions of the two groups in a disubstituted aromatic ring:ĭepending on the group (X) that is initially present on the benzene ring, the second substituent goes either to ortho/para or the meta position: In this post, we will talk about the ortho-, meta and para directors in electrophilic aromatic substitution (EAS). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
